Some of it is things you will have heard before, but its really just what I found best from first hand experience.
- Don't worry about them. The hardest thing to do, but the best: when you come out the other end, you will be thinking 'why did I even feel stressed!?'
- Sleep. Early nights, especially before exams.
- Treat yourself. Woo! For revising and during the exam period: I got through a lot of flavoured popcorn doing this blog.
- Revise.
- Practise. Do every past paper you can find.
- Teach someone else; the best way to learn is to explain to other people.
- Don't be stupid (go to hospital/do drugs before your exam.)
- Bring all the right equipment to the exam- you just feel so prepared and in control.
- Start revising really early to lighten the load and get things in you long term memory.
- Deep breathing, kept me calm, focused and stress free ;)

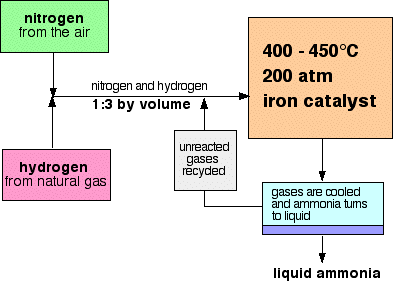



.gif)
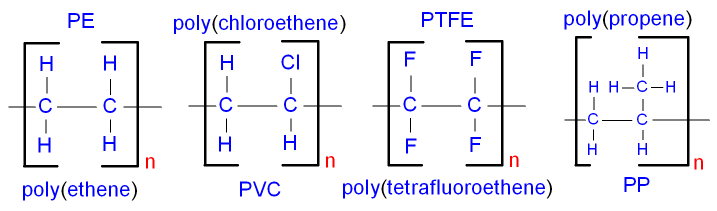

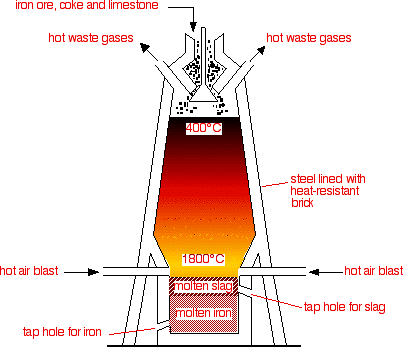



.jpg)

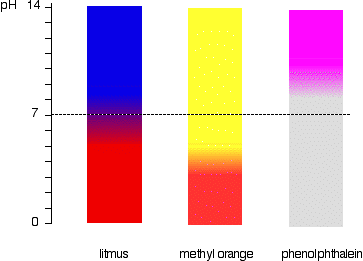
.jpg)
